
Molly Fruecht, 19, participates in a clinical trial at the University of Chicago to test a wearable device that monitors blood sugar and delivers the exact insulin dose the subject needs in order to get blood sugar into target range.
A team of University of Chicago Medicine, Illinois Institute of Technology (IIT), and University of Illinois-Chicago (UIC) researchers are developing an artificial pancreas that uses data from a wearable device and a glucose sensor to calculate and administer the exact dose of insulin that a person with Type 1 diabetes needs – in real time.
According to the CDC, about 30 million people in the U.S. have diabetes. Type 1 diabetes, where the body does not produce the hormone insulin, accounts for about 5 percent of adult cases of diabetes. Formerly known as juvenile diabetes, this condition results in too much sugar, or glucose, in the blood and requires that those who have it be dependent on insulin to control their blood sugar. They must monitor their glucose levels regularly and are at risk for serious related health issues, such as blindness, amputated limbs, and heart disease.

Elizabeth Littlejohn, MD
“An artificial pancreas system to manage insulin is the next kind of cure for this condition and takes technology for blood sugar monitoring to the next level,” said pediatric endocrinologist Elizabeth Littlejohn, MD, UChicago Associate Professor of Pediatrics and Medicine, who is leading the study at UChicago.
Thanks to early-stage funding from UChicago’s Diabetes Research and Training Center’s Pilot & Feasibility Studies program and specialized research facilities at the Institute of Translational Medicine’s (ITM) Clinical Research Center (CRC), the device is currently being used by patients and evaluated by the research team in a series of clinical trials at the ITM’s CRC.
The CRC has provided investigators with support for complex clinical and translational science projects for more than 56 years, offering skilled research nurses and staff, lab assistance for handling biological samples and assay performance, and exclusive space to perform their studies. Littlejohn and her collaborators study has gone on to receive more than $5 million in additional funding from the National Institutes of Health (NIH) and nearly $600,000 in funding from the Juvenile Diabetes Research Fund.

This is an example of the type of wearable device that captures data for the artificial pancreas. Image source: Turksoy K, Quinn LT, Littlejohn E, Cinar A.
The artificial pancreas system functions as three parts:
- Sensors (glucose sensors and the wearable devices) to take real-time measurements of glucose levels, skin moisture and energy expenditure
- A control system that determines the rate at which insulin is delivered to the patient to quickly regulate the level of glucose in the blood.
The heart of the technology lies in a complex algorithm that takes into account everything from patients’ heart rate to their glucose level and energy expenditure in order to calculate how much insulin they need.
It will even tell the user whether an insulin injection or eating carbohydrates, like whole grains or starchy vegetables, is the best way to get their blood sugar into range.

Ali Cinar, PhD
The artificial pancreas is a “smart” machine that runs on an adaptive predictive mode, which means it gathers information from the past readings to “learn” continuously from patients to predict the future, allowing it to develop safe patterns of blood sugar levels over time.
“Rather than just reacting to the present, these adaptive controllers can think about what will happen down the road and can adjust accordingly,” said Ali Cinar, PhD, Professor of Chemical Engineering and Director of the Engineering Center for Diabetes Research and Education at IIT, who is a collaborator on the project and lead principal investigator (PI).
The system also uses the patient’s biometric/metabolic information from the wearable devices to improve the accuracy of its predictions and determine the presence and intensity of physical activity. It requires no manual inputs from users, relieving them from the stress of counting carbs or estimating exercise effects.
Researchers from IIT, UChicago, and UIC came to collaborate on this project to harness their strengths in engineering expertise, data management, and clinical trials.
In recent weeks, thirteen clinical trial participants have tested the device at the ITM’s CRC.
During these tests, CRC staff connect a small athletic-style arm band, which communicates wirelessly with a laptop containing a controller that both monitors data and provides insulin suggestions.
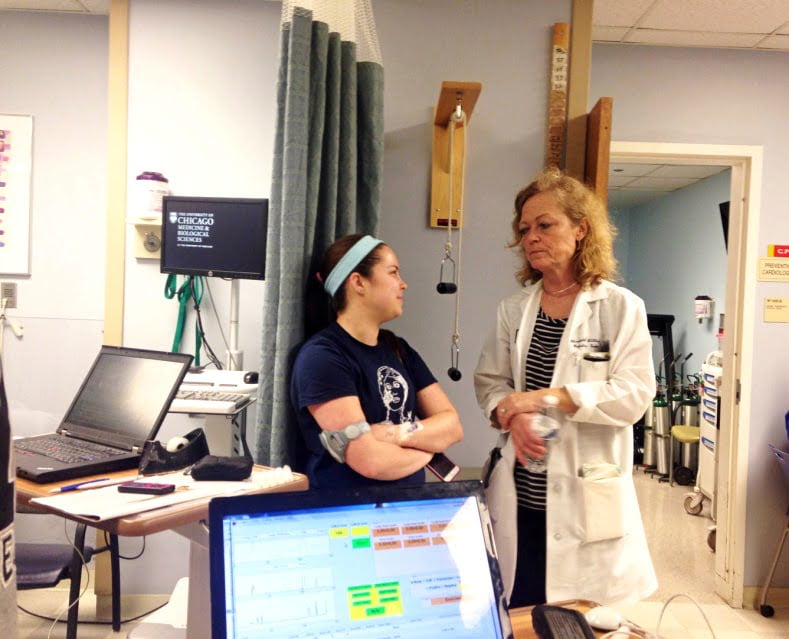
Elizabeth Littlejohn, MD, talks with clinical trial participant Molly Fruecht during one of Fruecht’s visits to the UChicago ITM Clinical Research Center (CRC).
With their arm bands on, participants sleep, eat, and exercise as the algorithm evaluates their information and recommends insulin doses along the way. The CRC’s Bionutrition Core provides weighed meals for each participant, calculating and tailoring the calories and other nutritional content to each person’s needs and preferences.
Participant Molly Fruecht, 19, is no stranger to the medical world — she has completed other clinical trials as a Type 1 diabetes participant before and is studying integrative biology at University of Illinois at Urbana-Champaign (UIUC). Her interest in biology overlaps with her interest in research — which is why she said she participates in clinical trials like the artificial pancreas project.
The technology is low-maintenance care for diabetic people like Fruecht.
“It’s really exciting because they get to deal with the diabetes, and I just get to sit here and watch Netflix,” Fruecht said.
Most people with diabetes must monitor their blood sugar levels and do so by pricking their fingers for a blood test on a glucose monitor. However, residue on their fingers, monitor errors, and other issues can cause inaccurate readings.
An artificial pancreas reduces the risk of this type of error by collecting data from a sensor underneath the skin. It does the work of automatically adjusting insulin for the subject’s needs at that moment.
“The device is going from a lab model to use on a human being, a moving, breathing, exercising person,” said Littlejohn, who is also the Associate Clinical Director of UChicago’s Kovler Diabetes Center. “It will then go beyond the CRC to further testing, and then hopefully, it will go home for patients to use.”

Laurie Quinn, PhD, RN
But before it can go to patients’ homes, the device has to become portable.
The algorithm currently is stored on a laptop, which means its need for a computer limits mobility. Littlejohn’s team hopes to put the algorithm into a cell phone that receives metabolic information and then instructs the pump what dose of insulin to give.
Physicians could also monitor all of their patients’ data remotely if that information is synced to a cloud storage program.
Within the next few years, Littlejohn said they want to take the device to a younger population, as diabetes is a common chronic disease among adolescents and children. She plans to take the arm bands and artificial pancreas to camps for children with diabetes, where medical personnel would be available to help monitor its use.
The research team, which includes Cinar and UIC’s Laurie Quinn, PhD, RN, have published their trial results in journals such as IEEE Transactions on Biomedical Engineering, Industrial & Engineering Chemistry Research, the Journal of Diabetes Science and Technology, and Diabetes Technology & Therapeutics.
To get involved or donate to the project, please contact Elizabeth Littlejohn, MD, at elittlej@peds.bsd.uchicago.edu.


